1. The Three Methods at a Glance
When it comes to measuring pH, you have three primary options: electronic pH meters, pH test strips (pH paper), and liquid pH indicators. Each method has distinct advantages and limitations in accuracy, cost, ease of use, and application suitability.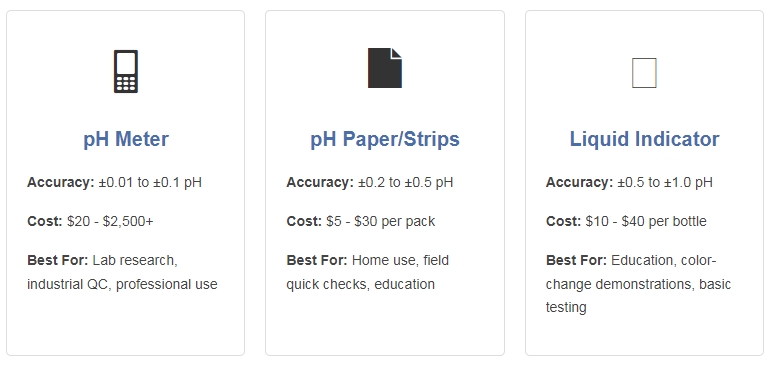
2. Detailed Method Comparison
| Feature | pH Meter | pH Paper/Strips | Liquid Indicator |
|---|---|---|---|
| Accuracy |
±0.01-0.1 pH (lab grade) ±0.1-0.2 pH (portable) |
±0.2-0.5 pH (wide range) ±0.1-0.2 pH (narrow range) |
±0.5-1.0 pH (depending on indicator) |
| Resolution |
0.01-0.1 pH显示); 0.001 pH (research) |
0.2-0.5 pH (color chart interval) | 0.5-1.0 pH (color transition) |
| Measurement Time | 15-60 seconds (stabilization) | 1-5 seconds (immediate) | 1-3 seconds (immediate) |
| Sample Size Needed | 5-50 mL (depends on electrode) | 0.5-1 mL (small drop) | 2-5 mL (enough to see color) |
| Temperature Compensation | Yes (ATC on most models) | No (temperature affects accuracy) | No (temperature affects accuracy) |
| Calibration Required | Yes (daily or weekly) | No (factory calibrated) | No (but indicator degrades over time) |
| Affected by Color/Turbidity | No (electrode unaffected) | Yes (dark/colored samples hard to read) | Yes (sample color interferes) |
| Subjective Interpretation | No (digital display) | Yes (color matching varies by person) | Yes (color perception varies) |
| Initial Cost |
$20-$150 (basic) $300-$2,500 (lab/professional) |
$5-$30 per pack (50-200 strips) | $10-$40 per bottle (50-100 tests) |
| Cost Per Test | <$0.01 (after initial investment) |
$0.05-$0.50 per strip
|
$0.10-$0.50 per test |
3. pH Meters: The Gold Standard
3.1 How It Works
A pH meter uses a glass electrode and reference electrode to measure the electrical potential generated by hydrogen ions. The meter converts this potential to a pH reading using the Nernst equation, with automatic temperature compensation ensuring accuracy across varying temperatures.
3.2 Advantages
- Highest accuracy: ±0.01 pH or better, 10-100× more precise than paper
- Digital readout: No subjective color interpretation
- Unaffected by sample color: Works with dark, turbid, or colored samples
- Data logging: Many models store measurements for record-keeping
- Low per-test cost: After initial investment, cost per test is negligible
3.3 Disadvantages
- Higher initial cost: $20 for basic, $300+ for professional
- Requires calibration: Needs regular calibration with buffer solutions
- Electrode maintenance: Must be stored properly and cleaned regularly
- Fragile: Glass electrode can break if mishandled
- Slower: Takes 15-60 seconds for reading to stabilize
Best For: Laboratories, quality control, research, industrial processes, professional water testing, and any application requiring high accuracy (±0.1 pH or better).
4. pH Paper/Test Strips: The Convenient Choice
4.1 How It Works
pH paper is impregnated with one or more indicator dyes that change color at specific pH values. When dipped in a solution, the paper changes color, and the user compares it to a printed color chart. Narrow-range strips offer higher accuracy for specific pH ranges.
4.2 Advantages
- Extremely affordable: Packs cost $5-30 for 50-200 tests
- No calibration needed: Ready to use out of the package
- Very fast: Results in 1-5 seconds
- Portable: Pocket-sized, no batteries or maintenance
- Disposable: No cleaning required—just discard after use
- Wide range available: pH 0-14 or narrow ranges (e.g., 6.0-7.6)
4.3 Disadvantages
- Lower accuracy: Typically ±0.2-0.5 pH (wide range) or ±0.1-0.2 (narrow range)
- Subjective interpretation: Color matching varies between users
- Affected by lighting: Different light sources change perceived color
- Not for colored samples: Dark or colored samples make reading difficult
- Temperature sensitive: Accuracy decreases outside 20-25°C
- Expiration: Strips degrade over time, especially if exposed to humidity
Best For: Home use (aquariums, pools, hydroponics), field quick checks, education (student labs), and any application where ±0.3-0.5 pH accuracy is sufficient.
4.4 Types of pH Paper
| Type | pH Range | Resolution | Best Use |
|---|---|---|---|
| Full range (universal) | 0-14 | 0.5-1.0 pH increments | Quick general screening |
| Narrow range | e.g., 3.0-5.5, 6.0-7.6, 7.5-9.5 | 0.2-0.3 pH increments | Specific applications (aquarium, pool, soil) |
| Precision narrow | e.g., 6.8-7.4 (blood/urine) | 0.1-0.2 pH increments | Medical/diagnostic applications |
| Litmus paper | 4.5-8.3 (red to blue) | 1-2 pH increments (acid/base only) | Simple acid/base discrimination only |
5. Liquid pH Indicators: The Classic Method
5.1 How It Works
Liquid indicators are dye solutions that change color over specific pH ranges. A few drops are added to the sample, and the resulting color is compared to a reference chart. Common indicators include phenolphthalein (colorless at pH 8.3, pink at pH 10.0) and universal indicator (multiple colors across pH 0-14).
5.2 Advantages
- Simple visual detection: No equipment needed
- Low cost: $10-40 per bottle (50-100 tests)
- No calibration required: Ready to use
- Good for demonstrations: Vivid color changes for teaching
- Specific indicators available: Target specific pH ranges
5.3 Disadvantages
- Lowest accuracy: ±0.5-1.0 pH (depends on indicator)
- Subjective interpretation: Color perception varies significantly
- Affected by sample color: Cannot use with colored or turbid samples
- Temperature sensitive: Color transition changes with temperature
- Indicator degrades: Solutions have limited shelf life
- Contaminates sample: Cannot reuse sample after adding indicator
5.4 Common Indicators and Their Ranges
| Indicator | pH Range | Color Change | Common Use |
|---|---|---|---|
| Methyl orange | 3.1-4.4 | Red → yellow | Acid titrations |
| Bromothymol blue | 6.0-7.6 | Yellow → blue | Aquarium, swimming pool |
| Phenolphthalein | 8.2-10.0 | Colorless → pink | Base titrations |
| Universal indicator | 0-14 | Multiple colors (red→yellow→green→blue→purple) | General pH estimation |
Best For: Educational demonstrations, acid/base titrations in chemistry labs, and applications where only approximate pH (±1.0) is needed.
6. How to Choose: Decision Flowchart
What accuracy do you need?
Lab/QC/Research
→ pH Meter
Home/Field/Screening
→ pH Paper
Education/Demos
→ Indicator
Selection Checklist:
7. Application-Specific Recommendations
| Application | Recommended Method | Reason |
|---|---|---|
| Laboratory research | pH meter (benchtop, ±0.01) | High accuracy, data logging, reproducibility required |
| Quality control (food/beverage/pharma) | pH meter (industrial/lab) | Regulatory compliance requires documented accuracy |
| Wastewater treatment | pH meter (online/portable) | Continuous monitoring, ±0.1 accuracy sufficient |
| Aquarium (freshwater) | pH paper (narrow range 6.0-7.6) or basic pH meter | Moderate accuracy needed; paper is cheaper for casual use |
| Swimming pool/spa | pH paper (wide range) or test kit with indicator | ±0.3 accuracy sufficient; cost-effective |
| Hydroponics | pH meter (portable, ±0.1) or narrow-range paper (5.5-6.5) | Plants need precise pH; meter recommended for serious growers |
| Soil testing | pH meter (with soil probe) or soil test kit | Specialized electrodes needed; paper less accurate with soil slurries |
| Home brewing / winemaking | pH meter (portable, ±0.1) or narrow-range paper | Critical for fermentation; meter recommended for consistency |
| Drinking water (home) | pH paper (wide range) or basic pH meter | ±0.3 accuracy is fine; paper is cheaper for occasional testing |
| Educational demonstrations | Liquid indicator or pH paper |
Visual color changes engage students; lower cost for class sets
|
8. Accuracy Comparison: Real-World Testing
In a controlled study comparing methods on standard buffer solutions:
- pH Meter (calibrated): Deviation < ±0.02 pH from true value
- Narrow-range pH paper: Average deviation ±0.15-0.25 pH (depending on user)
- Wide-range pH paper: Average deviation ±0.4-0.6 pH
- Universal indicator liquid: Average deviation ±0.6-1.0 pH
8.1 Color Perception Variability
Studies show that color interpretation varies significantly between individuals:
- Different users can report pH values differing by 0.3-0.5 pH using the same strip
- Lighting conditions (fluorescent, LED, daylight) change perceived color by 0.2-0.4 pH
- Color blindness affects approximately 8% of males and 0.5% of females
Key Takeaway: If your application requires consistent, objective results (regulatory reporting, quality control, research), a pH meter is essential. For quick, approximate checks where ±0.5 pH is acceptable, pH paper is sufficient.
9. Cost Analysis: Long-Term Ownership
| Cost Factor | pH Meter | pH Paper | Liquid Indicator | |
|---|---|---|---|---|
| Initial equipment |
$50-300 (basic portable) $500-2,500 (lab benchtop) |
$10-30 (pack of strips) | $15-40 (bottle of indicator) | |
| Per-test cost (ongoing) | ~$0.01 (calibration solutions, electrode replacement) |
$0.05-0.50 per strip
|
$0.10-0.50 per test
|
|
| Annual consumables (1000 tests) | $50-150 (buffers, storage solution, electrode care) | $50-300 (depending on strip quality) | $100-500 (depending on usage) | |
| Maintenance | Regular cleaning, storage, calibration, eventual electrode replacement ($50-200/year) | None (disposable) | None (but indicator degrades over time) | |
| 3-Year Total Cost (1000 tests/year) | $500-1,500 (depending on equipment quality) | $150-600 (depending on strip quality) | $300-1,500 (indicator + frequent replacement) |
For frequent testing (>500 tests/year), a pH meter becomes more cost-effective than paper despite higher initial investment. For occasional testing (<100 tests/year), pH paper is usually the most economical choice.
10. Frequently Asked Questions
Q: Is pH paper accurate enough for my aquarium?
A: Yes, for most freshwater aquariums, narrow-range pH paper (6.0-7.6) provides sufficient accuracy (±0.2-0.3 pH). For saltwater or reef tanks requiring precise control (±0.1 pH), a pH meter is recommended.
Q: Can I use pH paper with colored samples?
A: Not reliably. Dark or colored samples (wine, juice, colored solutions) interfere with color interpretation. Use a pH meter for colored samples.
Q: How long do pH strips last?
A: Unopened, 2-3 years. Once opened, 6-12 months if stored properly (cool, dry, dark). Discard if colors look faded or discolored.
Q: Do I need to calibrate a pH meter every time I use it?
A: For high accuracy, calibrate daily or before each use. For general use, calibrate weekly or when readings seem off. Check with pH 7 buffer to verify calibration.
Q: Which is better for home brewing: pH meter or paper?
A: A pH meter is recommended for serious brewing. Mash pH is critical (5.2-5.6 target range), and ±0.1 accuracy matters for enzyme activity and final beer quality.
Q: Can liquid indicators be used with turbid samples?
A: No. Suspended particles scatter light and make color interpretation impossible. Use a pH meter for turbid samples.
11. Summary: Quick Selection Guide
Choose a pH Meter If:
- You need accuracy of ±0.1 pH or better
- You test frequently (>200 tests/year)
- Your samples are colored, turbid, or opaque
- You need digital records or data logging
- Regulatory compliance requires documented accuracy
- You have budget for initial investment ($50+)
Choose pH Paper/Strips If:
- ±0.3-0.5 pH accuracy is sufficient
- You test occasionally (<100 tests/year)
- You need quick, on-the-spot results
- Your samples are clear and colorless
- You have limited budget ($10-30)
- You don't want calibration or maintenance
Choose Liquid Indicator If:
- You need a visual demonstration for teaching
- ±0.5-1.0 pH accuracy is acceptable
- You are performing acid/base titrations
- You have clear, colorless samples
- You want a classic chemistry experience
Final Recommendation: For professional, laboratory, or regulatory applications: pH Meter. For home/hobbyist applications with clear samples: Narrow-range pH paper. For teaching/demonstrations: Liquid indicators.